Key takeaways:
- Treating cancer based on only genomic mutations, and not the specific malignancy, does not show survival benefits.
- Many patients with evidence-based mutations do not receive targeted therapies.
Treatments based on genomic profiling can significantly extend survival for patients with advanced or rare cancer, but only if those therapies have been validated within their histologic type.
An evaluation of more than 3,000 adults in Australia showed those who received regulatory-approved targeted therapies or ones that have demonstrated clinical activity within a patient’s specific cancer had a 40% improvement in survival compared with individuals who did not receive those treatments.

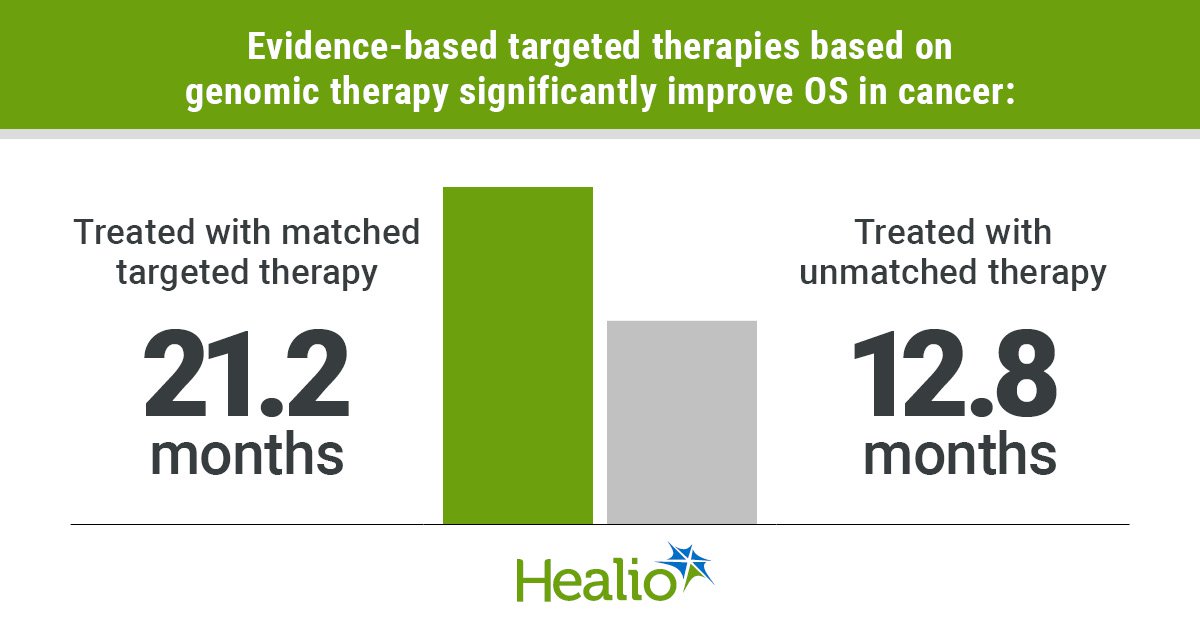
Data derived from Lin FP, et al. JAMA Oncol. 2026;doi:10.1001/jamaoncol.2026.0127.
However, individuals who received genomic treatments based only on data from a different malignancy derived no meaningful survival benefit.
“Treat the cancer, not just the mutation,” Frank P. Lin, MB ChB, PhD, medical oncologist and senior research fellow at NHMRC Clinical Trials Centre at University of Sydney, told Healio. “Based on our data, prescribing a drug because it works for a given mutation in another cancer type, without histotype-specific trial evidence, cannot be endorsed as standard practice at present. Our data suggest it does not show a difference in survival compared to unmatched therapy. In fact, the trend might be reversed.”
‘There’s a revolution going on’
Researchers in Australia developed the Molecular Screening and Therapeutic (MoST) program in 2015 to find targetable biomarkers for patients with advanced, neglected or rare cancers through genomic profiling, according to study background.

David M. Thomas
“It’s pretty clear there’s a revolution going on in cancer treatment,” David M. Thomas, MBBS, PhD, director of the Centre for Molecular Oncology at University of New South Wales, told Healio.
“In Australia, screening for biomarkers is not standard of care. For patients, and to assist in the execution of clinical trials of novel biomarker-dependent therapies, we set up a national screening program specifically designed to identify therapeutic biomarkers in patient populations in need of new treatments.”
Using the screening data from MoST, researchers evaluated use of targeted therapies and their impact on survival.
They used the Therapy-Oriented Precision Oncology Guidelines for Recommending Anticancer Pharmaceuticals (TOPOGRAPH) framework to tier medications patients received. The categories included:
- Tiers 1 and 2: Therapies approved based on a specific biomarker in a specific cancer
- Tier 3A: Therapies that showed clinical activity in a certain cancer but had not been approved
- Tier 3B: Therapies that demonstrated efficacy for a biomarker in one cancer but lacked data in the treated malignancy
- Tier 4: Therapies with preclinical evidence
- Tier R2: Therapies predicted to not have activity
The analysis included data from 3,383 adults (mean age, 57.1 years; standard deviation, 14.3; 53% women) with advanced or metastatic cancers who had exhausted all standard systemic therapeutic options and who underwent genomic profiling between June 2016 and December 2021.
The most common malignancies included sarcoma (19.3%), colorectal cancer (10.2%) and pancreatic cancer (8.5%). Lin, Thomas and colleagues classified 71.7% of participants with a “rare” cancer.
OS from the date of genomic profiling served as the primary endpoint.
“Precision oncology has long operated on an appealingly simple promise: identify the right mutation and find the right drug,” Lin said. “However, physicians working at the coalface, in the clinic and at molecular tumor board meetings, know the reality is considerably more complex. When a patient hands you a dense genomic report after exhausting standard options, the question is rarely whether a mutation is present. The harder question is, how strong is the clinical evidence that this specific drug will meaningfully alter the course of this specific cancer?”
‘That’s problematic’
Overall, 88.8% of patients had a tier 1 to 4 biomarker, including 37.5% with a tier 1 to 3A target. However, only 39% of the study population received additional therapy based on genomic profiling.
“That’s problematic,” Thomas said. “It doesn’t relate to the science. It relates to the availability of drugs, either through reimbursed access or through clinical trials. Frankly, we’ve got a bit of work to do to increase the number of treatment options that these patients can access.”
After median follow-up of 22.6 months, 63% of the cohort died.
The study population had a median OS of 11.3 months, but patients who received additional genomic-based care had substantially longer survival (median OS, 14.1 months vs. 8.2 months).
Among patients who did not receive additional therapy, those who had a tier 1 to 4 target had significantly shorter survival than those without a matching target (median OS, 7.1 months vs. 30.1 months; adjusted HR = 1.75; 95% CI, 1.38-2.21).
Among patients who received additional therapy, 29% received treatment matched to tier 1 to 4 biomarkers. Those who had a tier 1 to 3A biomarker and got matched treatment had significantly longer OS than those who received unmatched therapy (21.2 months vs. 12.8 months; aHR = 0.6; 95% CI, 0.44-0.82). Patients who had a tier 3B to 4 biomarker and received matched therapy had similar survival as those who received unmatched care (14.5 months vs. 12.8 months). Among those who had a tier 3B biomarker specifically, treatment with matched therapy did not result in significantly longer OS, either.
“Biological context matters immensely in drug selection,” Lin said. “A classic example is the BRAF V600 mutation. When found in melanoma, it is highly responsive to targeted treatment, but that same mutation in colorectal cancer behaves entirely differently and requires a completely different treatment paradigm. As oncologists working in the clinic, we should reconsider the appropriateness of reflexive molecular drug repurposing and instead direct patients toward appropriately designed clinical trials.”
Researchers acknowledged study limitations, including use of TOPOGRAPH for tiering medications.
“While our study provides compelling observational evidence that the TOPOGRAPH tiering system stratifies clinical outcomes in a meaningful way, definitive prospective and comparative evaluation is needed to compare it with other frameworks, such as European Society for Medical Oncology Scale for Clinical Actionability of Molecular Targets,” Lin said. “As an oncology community, we need to work together to understand the best strategy for moving forward to underpin clinical decision-making at scale.”
‘We need to increase access’
Both Lin and Thomas highlighted the importance of improving access to matched treatments in the future.
“Overall, 39% of patients [in our study] had a treatment recommendation which we show would extend survival, but only 3.4% of that group, 1 in 10, actually went on to receive treatment,” Thomas said. “At the very least, one would expect that every patient who is dying of cancer who carries a biomarker that would extend survival would have an option to access those treatments.”
Thomas noted only 8% of patients with cancer in Australia currently participate in clinical trials.
“We need to increase access to clinical trials, and for the drugs that are at the right stage, those drugs need to pass through our regulatory process so they do become a standard of care,” he said.
Identifying barriers to treatments could increase uptake of matched therapies.
“Whether [low uptake] is due to clinical ineligibility, drug access, regulatory hurdles, reimbursement issues or patient preference needs to be further studied,” Lin said. “Given the limitations of our data, which extend back to 2021, we also need to continue to examine if those dynamics have evolved over the study period as access has improved.”
Because the field is “constantly moving,” systems need to be refined so clinicians have the most updated information on who should receive matched genomic therapies, Thomas said.
“Since that trial was conducted, the same programs have continued, and currently our recommendation rate is not 65%, it’s 85%, and the actionability rate that will extend survival has jumped from 39% to 45%,” Thomas explained. “I’m expecting that trend to continue. In fact. I’m expecting that by 2035, every patient that we screen should have a biomarker that would predict at least that there is a matched drug, if not an excellent match drug, and at least half of those patients will have a drug available that will double their survival.”
“The future of genomic medicine in oncology does not rest primarily on sequencing technology becoming cheaper or faster,” Lin added. “Rather, it rests on building the clinical trial ecosystems, the international data-sharing infrastructure, and the decision-support frameworks that allow us to convert genomic information into durable, evidence-based patient benefit. That will be as much a global scientific and institutional challenge as a technological one.”
For more information:
Frank P. Lin, MB ChB, PhD, can be reached at frank.lin@sydney.edu.au.
David M. Thomas, MBBS, PhD, can be reached at dmthomas@unsw.edu.au.
<















Leave a Reply