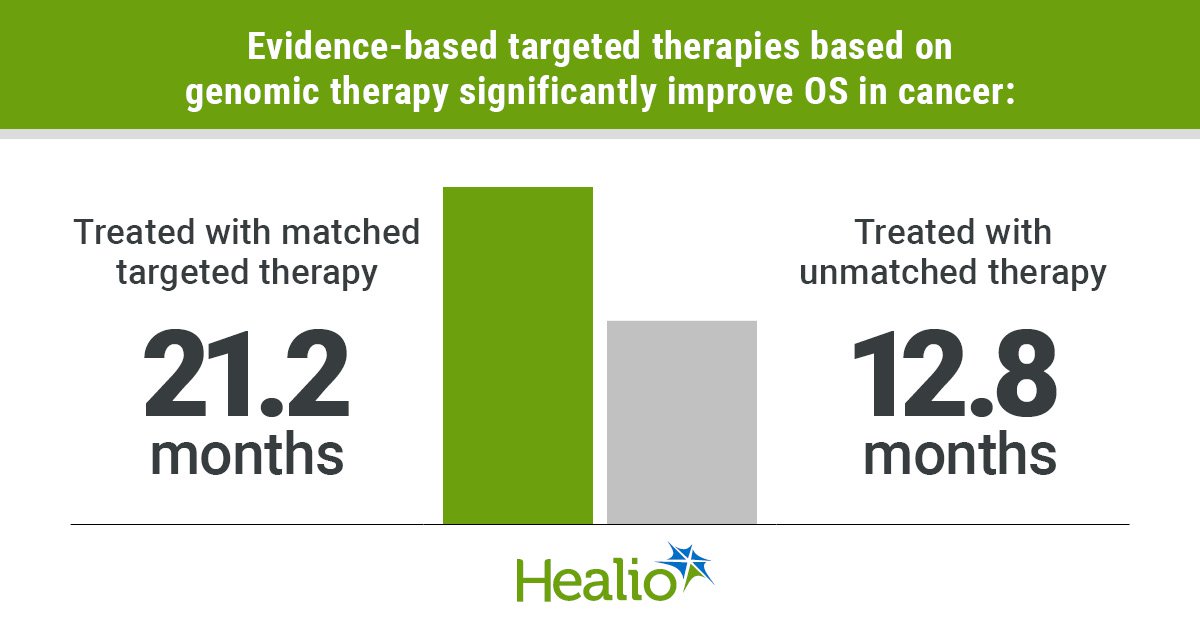
Key takeaways:
- The FDA authorized the marketing of four non-tobacco and non-menthol fruit-flavored vapes.
- The vapes require age verification that is “expected to effectively mitigate” usage among youth.
The FDA for the first time authorized the marketing of non-tobacco and non-menthol electronic nicotine delivery system products for adults, the agency announced.
The four vapes authorized via the FDA’s premarket tobacco product application pathway — which come in gold, sapphire, classic menthol and fresh menthol flavors — contain 50 mg/mL, or 5%, of tobacco-derived nicotine, according to an FDA press release.

The FDA authorized the marketing of four non-tobacco and non-menthol fruit-flavored vapes. Image: Adobe Stock
“By helping to prevent youth use, device access restrictions are a potential game changer,” Bret Koplow, PhD, JD, acting director of the FDA’s Center for Tobacco Products, said in the release. “This technology is also an indication of the role innovation may serve in the effort to protect young people from threats posed by nicotine use and addiction while helping to enable availability of an expanded array of flavored options for adults who smoke who may use these products to completely switch away from regular cigarettes.”
The FDA said that its “rigorous, scientific review” of the products manufactured by Glas, Inc. “found that the applicant sufficiently demonstrated that Glas’s device access restriction technology, combined with FDA-required marketing restrictions, is expected to effectively mitigate the ability of youth to use the product.”
The manufacturer’s technology “requires the user to verify their age and identity with a government-issued ID and pair the device with a smartphone via Bluetooth,” the release said. “Following verification, the device will not operate if separated from the phone. The app also conducts random biometric check-ins to periodically confirm the registered user is the one using the device.”
The FDA also said that Glas was able to demonstrate that most adults aged 21 years or older successfully verified their age and found the vapes’ instructions and age verification software easily understandable “while youth and young adults could not.”
“With these orders, the FDA confirms the availability of an additional way for demonstrating that a non-tobacco flavored product meets the public health standard by using effective age-gating,” the release said. “It remains the case that a non-tobacco flavored product may be authorized where the application demonstrates its benefit in helping adults quit cigarette smoking outweighs its potential risks.”
E-cigarettes are usually less harmful vs. cigarettes, the FDA previously told Healio. However, e-cigarettes have been tied to several conditions, including elevated BP and hypertension, peptic ulcer disease, lung cancer and asthma.
The FDA said the authorization requires Glas to ensure the vapes’ advertising, promotion and marketing is “carefully targeted” at adults aged 21 years or older while also tracking, measuring and reporting to the FDA the effectiveness of its prevention measures aimed at youth.
The agency said it could suspend or withdraw its authorization if the vapes’ marketing is “is no longer appropriate for the protection of the public health,” which could include a rise in usage among youth or if the products’ benefits no longer outweigh their risks.
Steven Kelder, PhD, MPH, a professor of epidemiology at the University of Texas Health Houston School of Public Health, told Healio that “one of things that’s very clear is that teens like flavors. That’s one of the primary causes about why they’re curious about them, and when they’re curious, they try them, and when they try them enough times, they get addicted. That’s my concern.”
“I’ve been hearing for decades now that adults need fruit flavors or they’re just not going to use vapes. The evidence doesn’t support that. If someone’s addicted to nicotine, they’re going to use nicotine,” Kelder said. “Physicians should know that there are risks in using vapes in terms of CVD and cancer. Vaping is less harmful, there’s no doubt about it, but they’re not harmless and the biggest harms fall on the kids. Why take a risk on these new flavors when its unnecessary.”
<















Leave a Reply