April 20, 2026
4 min read
Key takeaways:
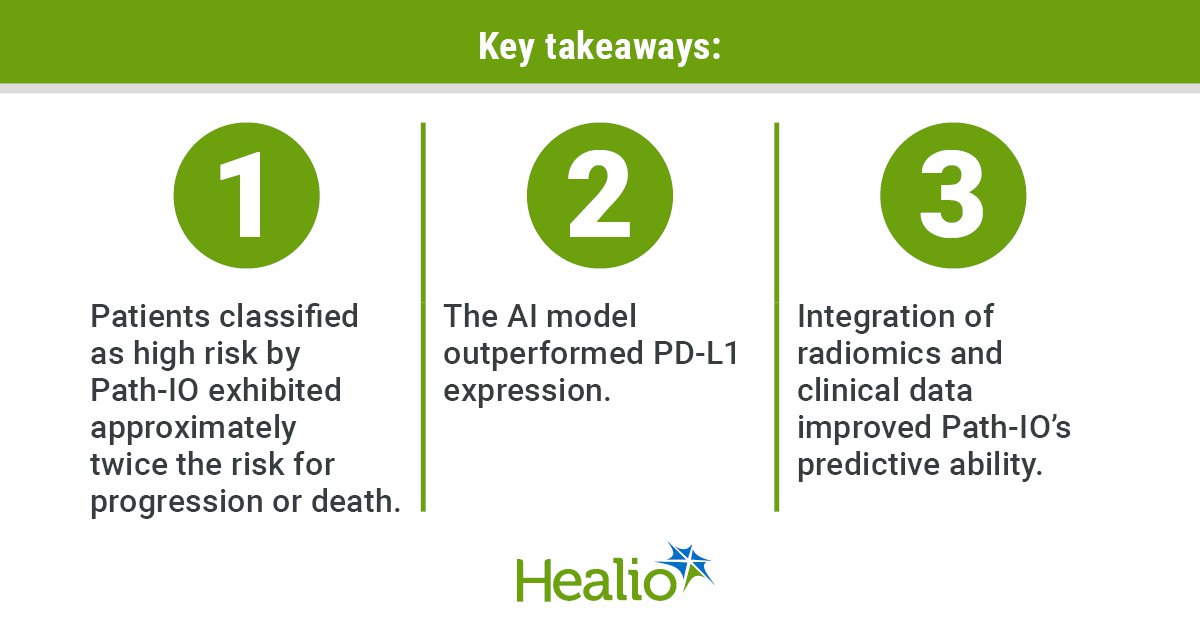
- A deep-learning approach predicted immunotherapy response in lung cancer better than PD-L1 expression, the standard biomarker.
- Integration of clinical data improved the model’s predictive ability.
A deep learning platform may help predict how people with advanced lung cancer will respond to immunotherapy, according to study results.
Patients determined by a biology-guided AI model to be at high risk for poor outcomes exhibited more than twice the risk for disease progression or death after immune checkpoint inhibitor therapy than those deemed to be at low risk, findings presented at American Association for Cancer Research Annual Meeting showed.

Data derived from Bandyopadhyay R, et al. Abstract 4003. Presented at: American Association for Cancer Research Annual Meeting; April 17-22, 2026; San Diego.

Rukhmini Bandyopadhyay
“Not all patients benefit from immunotherapy,” lead author Rukhmini Bandyopadhyay, PhD, postdoctoral fellow at The University of Texas MD Anderson Cancer Center, told Healio. “If we can identify which patients are most likely to benefit, we can give them a simpler treatment — maybe a monotherapy — that could result in fewer adverse effects. Those identified to be at higher risk [for poor outcomes] may need combination therapy, such as immunotherapy with chemotherapy, so this is very important for treatment decision-making.”
‘Google map’ for tumors
Immune checkpoint inhibitors have greatly improved outcomes for some individuals with cancer, with the most success observed in lung cancer, melanoma and genitourinary malignancies. However, approximately 80% of people with advanced cancer who receive these agents do not derive benefit.
Biomarkers used to predict response — such as PD-L1 expression or tumor mutational burden — are inconsistent and can be expensive, Bandyopadhyay said.
Pathomics — an emerging discipline in which AI is used to quantitatively analyze digital pathology reports to guide diagnosis and prognosis — could improve risk stratification.
Bandyopadhyay and colleagues developed a deep learning-based pathomics framework called Path-IO, the acronym for which stands for Pathology-driven Immunotherapy Optimization.
The AI model is designed to analyze hematoxylin/eosin-stained pathology slides to identify features within the tumor microenvironment and patterns across tissue samples, then predict whether a person has a lower or higher risk for poor outcomes with immunotherapy.
These slides are routinely used in clinical practice but even expert pathologists can have difficulty fully gleaning and understanding the high volume of complex data they contain, Bandyopadhyay said.
“You can think of a whole slide image as a Google map,” she said. “You have to zoom in to see different aspects of the tumor microenvironment or different tissue regions to see if there are tumor cells or immune cells to fight those tumor cells. This AI tool can help examine each of these small patches from the whole image to simplify this analysis.”
Biology-informed model
Bandyopadhyay and colleagues evaluated the model among patients with metastatic non-small cell lung cancer.
The analysis included 797 patients with NSCLC treated with immune checkpoint inhibitors at MD Anderson.
An external validation cohort included another 280 patients treated at two other institutions — Mayo Clinic and Gustave Roussy — or as part of a phase 3 trial that included immunotherapy-naive patients who received the anti-PD-1 antibody nivolumab (Opdivo, Bristol Myers Squibb) with or without the anti-CTLA-4 antibody ipilimumab (Yervoy, Bristol Myers Squibb).
Path-IO effectively stratified patients into higher-risk or lower-risk groups, with higher risk scores matching phenotypes that typically are not as sensitive to immunotherapy.
Patients classified as high risk exhibited approximately twice the risk for disease progression or death than those classified as low risk.
In the cohort of patients treated at MD Anderson, researchers reported HRs for PFS of 2.34 (P < .001) in the discovery set and 1.87 (P < .001) in the validation set, and HRs for OS of 2.11 (P < .001) in the discovery set and 2.51 (P < .001) in the validation set.
The model exhibited similar predictive potential in the validation cohorts at Mayo Clinic (HR for PFS = 2.45, P = .027; HR for OS = 2.46; P = .007) and Gustave Roussy (HR for PFS = 1.51, P = .046; HR for OS = 1.97, P = .003), as well as in the phase 3 trial cohort (HR for PFS = 2.76, P = .006; HR for OS = 1.78; P = .016).
Additional analyses in which researchers used the concordance index (C-index) to assess how well specific biomarkers distinguish patients with different outcomes showed Path-IO outperformed PD-L1 expression, the standard biomarker that guides use of immune checkpoint inhibitors. In the test cohort, PD-L1 alone achieved C-indices of 0.51 for PFS and 0.5 for OS. In contrast, Path-IO achieved C-indices of 0.58 for PFS and 0.63 for OS.
Researchers observed correlation between high risk scores via Path-IO assessment and immunologically “cold” phenotypes as determined by two other transcriptomics approaches.
The integration of radiomics and clinical data improved Path-IO’s predictive ability, increasing C-indices to 0.7 for PFS and 0.75 for OS. This finding underscores the importance of using multiple information sources during treatment decision-making, Bandyopadhyay said.
“The whole slide image provides tumor details at the cell level, CT images provide information at the macro level, and clinical factors like age, gender and whether cancer has metastasized provide additional information,” she said. “All of this information combined boosts the model’s ability to perform better.”
Prospective validation of the model is necessary, as is the integration of more comprehensive molecular profiling in hopes of further improving its predictive potential, Bandyopadhyay said. If validated, it would be simple and cost-effective to incorporate in routine practice given the pathology slides on which it relies are already widely used, she added.
“Prior studies in this area have used a mostly data-driven approach, and sometimes it is not clear why certain patterns are observed,” Bandyopadhyay said. “We are using a biology-informed model, which helps us understand why it is performing in such a robust way.”
For more information:
Rukhmini Bandyopadhyay, PhD, can be reached at rbandyopadhyay2@mdanderson.org.
<











Leave a Reply