The muscle damage that will weaken you at 60 may begin decades earlier.
Not as soreness or fatigue, but as a microscopic cascade you cannot feel: damaged proteins piling up, calcium pumps seizing, the molecular machinery of muscle contraction slowly breaking down — all while you go about your day, unaware.
Two companion studies recently published in Nature Metabolism have traced this invisible timeline, revealing that sarcopenia — the progressive muscle wasting that accelerates after 60 — doesn’t start when weakness appears. It starts years earlier, when a cellular cleanup system called chaperone-mediated autophagy (CMA) begins to fail.
“By midlife, you start losing CMA in the muscle,” said Ana Maria Cuervo, MD, PhD, distinguished professor of developmental and molecular biology at the Albert Einstein College of Medicine in Bronx, New York. She led the studies with collaborators at New York University, the National Institute on Aging, the Reynolds Oklahoma Center on Aging, and the Altos Labs San Diego Institute of Science.

These studies encourage reevaluation of the processes of sarcopenia and muscle aging because they also reveal something unexpected: a potential pathway to stalling and even reversing this decline.
The First Clues
Cuervo has spent more than three decades building the story of CMA, one organ at a time. From the liver she moved to the brain, linking the pathway’s failure to protein tangles in Alzheimer’s disease and alpha-synuclein aggregates in Parkinson’s disease, and showing how the pathway’s activation protects against retinal degeneration.
Each study added another tile to a mosaic of age-related decline, all tracing back to the same system.
Muscle was an unexplored frontier. When a postdoc with muscle expertise — Olaya Santiago-Fernández, PhD — joined Cuervo’s lab at the Albert Einstein College of Medicine to continue research started by Luisa Coletto, PhD, on the effects of CMA in muscle, the work intensified.

“I inherited this project from [Luisa],” Santiago-Fernández said. “I thought it had a lot of possibilities because the lab had already seen that when you block CMA specifically in skeletal muscle, you start to see muscle damage.” With her background in muscle biology, Santiago-Fernández was positioned to dig deeper into what that damage meant.
As the lead of one of the two companion studies, she and the team genetically engineered mice to lack CMA in their muscles. During strength tests, the mice showed a weak grip, and their endurance flagged on treadmill exercises. When the team dissected their muscles, the real story emerged.
“One of the main characteristics of muscle damage is that the cells’ nuclei, which are normally in the periphery of the fibers, relocate to the center,” Santiago-Fernández explained. In healthy muscles, nuclei hug the edges, leaving the interior packed with contractile machinery. But when they drift inward, it’s a sign that machinery has broken down.
“We also saw necrotic [dying tissue] areas, and that’s where we started to see this phenotype,” she said. “From there, we moved on to electron microscopy and saw more alterations.”
When CMA fails, damaged proteins accumulate and gum up the machinery of movement. But how does CMA work? Unlike bulk autophagy, which engulfs large swaths of cellular material wholesale to clean out debris and repurpose them into raw materials, CMA targets specific proteins bearing a particular molecular tag that marks them for disposal.
Chaperone proteins recognize this tag, escort the damaged protein to the lysosome, the cell’s recycling center, and thread it through a receptor called LAMP2A for degradation, hence the term “chaperone-mediated autophagy.”
But CMA doesn’t just eliminate garbage. The amino acids from the disposed proteins are released and become raw materials for building new, functional proteins. It’s a cleanup crew and supply chain in one.
Cuervo often explains the system with an analogy: “Imagine you go up a mountain in the cold because you have a house there, but the heat is not working. You see that you have a fireplace. You’re going to look around and say, ‘What do I have here that is old and that I can burn for fuel?’ Cells do the same.”
Seeing the Damage
When electron microscopy images of the damaged muscle in CMA-deficient mice came back, Cuervo was astonished by what she saw.
“You have all these beautiful muscle fibers, with everything aligned [in normal muscle],” Cuervo said, describing what healthy muscle looks like under an electron microscope.
There are dark bands called Z-lines on the ends of sarcomeres, the basic functional unit of muscle fibers responsible for contraction. Z-lines anchor the more central contractile machinery in the sarcomere as they stack in precise parallel rows like rungs on a ladder.
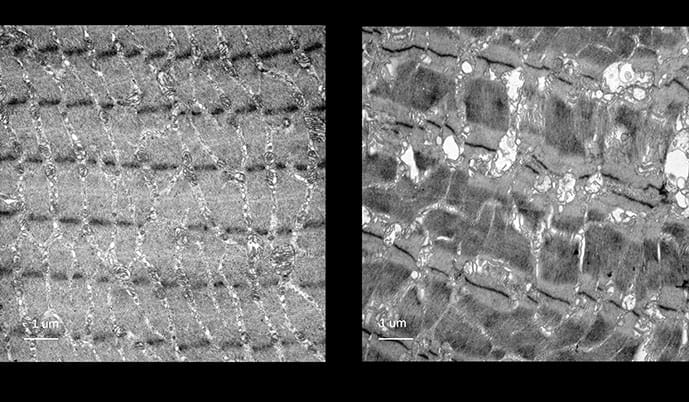
“But you look at these other muscles [in mice with CMA removed], and all this alignment [in the sarcomere] is lost.”
In the CMA-deficient mice, the Z-lines were distorted and scattered, with the orderly architecture of contraction in disarray. Even the mitochondria had swollen into dysfunctional-looking balloons. The membrane network responsible for calcium storage, the sarcoplasmic reticulum (SR), was also severely disrupted.
“That was the aha moment,” Cuervo said. “I kept showing that image to all the collaborators to convince them, and they said, ‘We’ve never seen damage like this so severe.’”

Susmita Kaushik, PhD, co-author of the studies, describes the chills she experienced viewing those images. “I have goosebumps just speaking about it. It was a night and day difference. [I thought] How are these mice even functional?’”
Tracking the Damage to Its Source
When researchers measured which proteins were accumulating in the damaged muscle, one stood out: the sarcoplasmic and endoplasmic reticulum Ca²⁺ ATPase, or SERCA, a calcium pump critical for muscle contraction and relaxation.
During muscle contraction, calcium floods into the cell, activating receptors on the SR, which then releases most of the calcium needed for contraction. SERCA’s job is to rapidly pump most of it back into the SR, allowing the muscle to relax.
But SERCA bunched up into misfolded aggregates in mice without CMA.
“We first saw that SERCA levels were elevated in mice [without CMA],” Santiago-Fernández explained. “SERCA started to accumulate in a misfolded fashion, creating these big, toxic accumulations.”
“If SERCA is aggregating, calcium levels can’t go down,” Cuervo added. “Calcium levels increase, you contract the muscle, but then the cell has to put it away in storage. That’s what SERCA does.”
What They Saw in Humans
To validate the findings in people, Santiago-Fernández collaborated with geriatrician Luigi Ferrucci, MD, PhD, at the National Institute on Aging, who has access to large datasets of muscle samples from young and old human populations.
Even among healthy older adults without overt frailty, CMA was already declining. And in patients with sarcopenia, the decrease was dramatic. The molecular changes preceded functional decline, suggesting reduced CMA in muscle could serve as an early warning marker.
The muscle findings fit into Cuervo’s larger vision of aging as a systemic failure of cellular maintenance.
“You don’t only age in one organ,” Cuervo said. “CMA is required in all the cells of your body. In the brain, as CMA declines, you will experience problems with memory and motor coordination. In the skeletal muscle, you have problems with force and movement. In the liver, you have problems with detoxification. In the retina, you have problems with sight.”
This systemic perspective shapes her therapeutic ambitions. The small molecules her lab has developed can even cross the blood-brain barrier, meaning they reach multiple organ systems. “By restoring CMA in many of the organs, you will have a more functional overall organism.”
The Double Hit: When Repair Fails Too
A companion study — conducted in collaboration with muscle stem cell expert Pura Muñoz-Cánoves, PhD, principal investigator at the Altos Labs San Diego Institute of Science — revealed an equally troubling finding about muscle regeneration.
Skeletal muscle fibers can’t divide to repair themselves. Instead, the body relies on satellite cells, a population of stem cells that sit dormant at the edges of muscle fibers. When injury occurs these stem cells activate, proliferate, and differentiate to rebuild damaged tissue. It’s a regenerative capacity that tissues such as the heart and brain largely lack.
In the study, mice lacking CMA in these stem cells couldn’t regenerate muscle. The researchers identified 723 proteins that CMA normally clears in these cells — all of which accumulate when the pathway fails. Glycolytic enzymes piled up in inactive forms, creating an energy crisis. And a protein called ARPC1B, which helps control the internal scaffolding that stem cells need to divide and move, accumulated abnormally.
The critical finding: Aged muscle stem cells — from both old mice and elderly humans — displayed the same defects as genetically CMA-deficient young cells. The pathway that fails with experimental deletion is the same one that fails naturally with aging.
“It’s not only that you can’t maintain your muscle,” Cuervo said, “but if you need to regenerate it because of damage, you can’t because now those cells are compromised.”
This creates what she calls a “double hit” — aging simultaneously undermines both the maintenance of existing muscle and the capacity to repair damage.
Reversal and Rescue
The therapeutic implications emerged from rescue experiments in both studies.
In the fiber study, researchers engineered mice to maintain LAMP2A expression throughout life and prevent the age-related decline because the loss of LAMP2A is the primary bottleneck in aging cells; by keeping this “entry gate” open, they allowed the continuous flow of cellular waste into the lysosome for degradation.
These animals preserved grip strength and muscle structure into extreme old age, which is the mouse equivalent of 95 human years.
In the stem cell study, pharmacologic activation proved equally effective. A small-molecule compound, which boosts CMA activity by inhibiting a natural CMA suppressor, restored the proliferative capacity of aged muscle stem cells.
Perhaps most intriguing was a metabolic workaround. Supplementing cells with pyruvate — a fuel that feeds into the cell’s energy machinery at a later stage, bypassing the broken enzymes — rescued proliferation even without fixing CMA itself.
Old human muscle cells responded similarly. Myoblasts from elderly donors, which showed impaired motility characteristic of CMA decline, regained their ability to move following CMA activation.
“Just a little bit of an increase [in CMA] is more than enough to boost the system for healthy aging,” Kaushik said.
Cuervo’s lab has spent 7 years developing CMA-activating compounds in collaboration with Evripidis Gavathiotis, PhD, medicinal chemist at Albert Einstein College of Medicine. “We gave them as pills,” she noted. “We don’t even have to inject the animals.” Einstein holds patents on these molecules, and licensing discussions with pharmaceutical companies are underway.
The studies also revealed that everyday choices influence CMA activity. Aerobic exercise such as running on the treadmill significantly activated CMA in mice skeletal muscle, and moderate exercise 30 minutes a day should be enough to activate it at least once daily.
Fasting works through similar logic: When dietary amino acids aren’t available, cells must recycle existing proteins to build new ones.
Monitoring Healthy Aging as We Get Older
Cuervo envisions a future where CMA is monitored as routinely as cholesterol, where markers for CMA can be measured like any other test. Catch the decline early, intervene before the damage accumulates, and perhaps the weakness that seems inevitable in old age won’t be inevitable at all.
But significant questions remain. Mouse models don’t perfectly replicate human aging. And the long-term effects of pharmacologically boosting CMA in humans are unknown. Though they’ve found that falling LAMP2A levels in aging mice trigger muscle decline, her lab is still working to find if additional factors contribute to CMA decline in humans.
But for the millions of older adults who have watched their strength slip away, who have struggled to rise from a chair or recover from a fall, these studies offer something that wasn’t there before: a target. Not just an explanation for why the body fails, but a mechanism that might be repaired.
The cleanup crew, it turns out, can be called back to work.
Cuervo was a co-founder and scientific advisor for the autophagy program at Life Biosciences during part of this project. Financial support was not provided for this work, and no data were shared with Life Biosciences, CalciMedica, or Altos Labs.
<













Leave a Reply