January 23, 2026
2 min read
Key takeaways:
- Researchers compared the outcomes of more than 40,000 infants who were immunized through nirsevimab or maternal RSV vaccination.
- Nirsevimab was associated with fewer severe outcomes than the maternal vaccine.
Nirsevimab may offer infants stronger protection against severe respiratory syncytial virus than maternal vaccination, real-world data suggests.
Many studies have shown that both the maternal RSV vaccine (Abrysvo, Pfizer) and nirsevimab (Beyfortus, AstraZeneca and Sanofi) are effective at reducing infants’ odds for severe RSV. The new study is the first to compare the two.

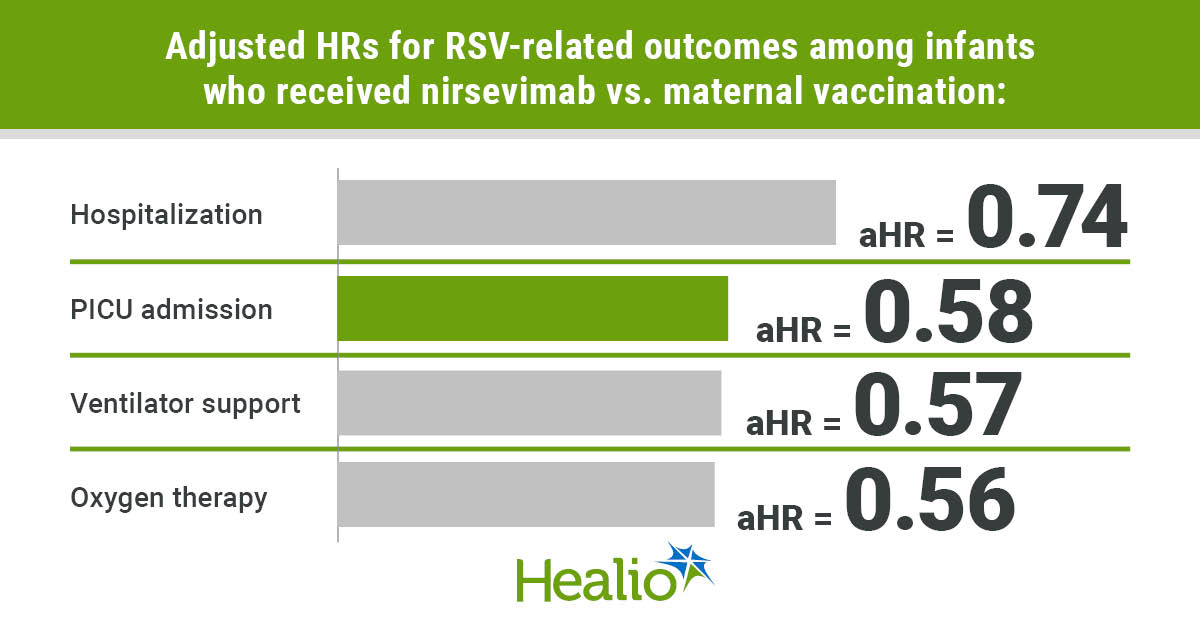
Data derived from Jabagi MJ, et al. JAMA. 2025;doi:10.1001/jama.2025.24082.
“What surprised us most was the magnitude and consistency of the difference we observed in real-world conditions,” Marie Joelle Jabagi, PharmD, PhD, MPH, an epidemiologist at the French National Agency for Medicines and Health Products Safety and French National Health Insurance, told Healio. “Despite maternal RSVpreF vaccination being highly efficacious in trials, passive infant immunization with nirsevimab was associated with a consistently lower risk of RSV-related hospitalization and severe outcomes across most analyses.”
The population-based cohort study included real-world data from 42,560 infants (51.7% boys; mean age, 3.7 days; standard deviation [SD], 1.4 days) who were born in France between Sept. 1, 2024, and Dec. 31, 2024. The researchers matched 21,280 infants who were exposed to the maternal RSV vaccine with 21,280 infants who received nirsevimab to compare each immunization’s effectiveness at preventing RSV-related complications. The median follow-up was 84 days (interquartile range, 70-99 days).
Overall, 481 infants (mean age, 38.9 days; SD, 22.2 days) were hospitalized for RSV-related lower respiratory tract infection (LRTI). Of these, 212 (44.1%) had received nirsevimab and 269 (55.9%) were exposed to the maternal RSV vaccine.
According to Jabagi and colleagues, infants who received nirsevimab had a significantly lower risk for medically attended LRTI than those who were protected through maternal vaccination (adjusted HR = 0.74; 95% CI, 0.61-0.88). Infants who received nirsevimab were also less likely to be admitted to the pediatric ICU (aHR = 0.58; 95% CI, 0.42-0.8), need oxygen therapy (HR = 0.56; 95% CI, 0.38-0.81) or require a ventilator (HR = 0.57; 95% CI, 0.4-0.81). The researchers reported similar results in subgroup and sensitivity analyses.
Notably, infants in the nirsevimab group experienced a drastic decrease in risk for LRTI over time, according to the authors. During the first 7 days after enrollment, the risk for LRTI was nearly three times higher for infants in the nirsevimab group vs. the maternal vaccination group (aHR = 2.94; 95% CI, 1.19-7.69), however their risk was only half that of the maternal vaccination group after 60 days (aHR = 0.51; 95% CI, 0.3-0.86).
Both products are still relatively new — nirsevimab and the maternal vaccine were first implemented during the 2023-2024 respiratory season — and Jabagi said it will take several seasons’ worth of data to evaluate the products’ performance in real-world settings.
“Our results should not be interpreted as evidence against maternal RSV vaccination,” Jabagi said. “Instead, they underscore that clinicians should individualize prevention strategies based on clinical context, access to care and timing within the RSV season. Both approaches remain valuable and may be complementary, particularly in efforts to maximize population-level protection against RSV.”
For more information:
Marie-Joelle Jabagi, PharmD, PhD, MPH, can be reached at pediatrics@healio.com.
<











Leave a Reply