During her lifetime, the beloved American actor Mary Tyler Moore was an outspoken advocate for type 1 diabetes (T1D) research. Since her passing in 2017, her husband, S. Robert Levine, MD, has led an ambitious initiative aimed at transforming the clinical approach to the diabetic retinal disease (DRD) that plagued Moore in her later years.
The initiative was first launched in 2018 in collaboration with the Juvenile Diabetes Research Foundation (now Breakthrough T1D), for which Moore had served as international chairperson for over 30 years. Initially called the “Restoring Vision Moonshot,” it was renamed the Mary Tyler Moore (MTM) Vision Initiative in 2022.
“Vision loss was the diabetes’ complication that affected Mary’s life the most. In her heart, Mary was a dancer. It was in dance that she found her joy. Diabetes stole her joy because diabetic retinal disease stole her vision,” Levine told Medscape Medical News.
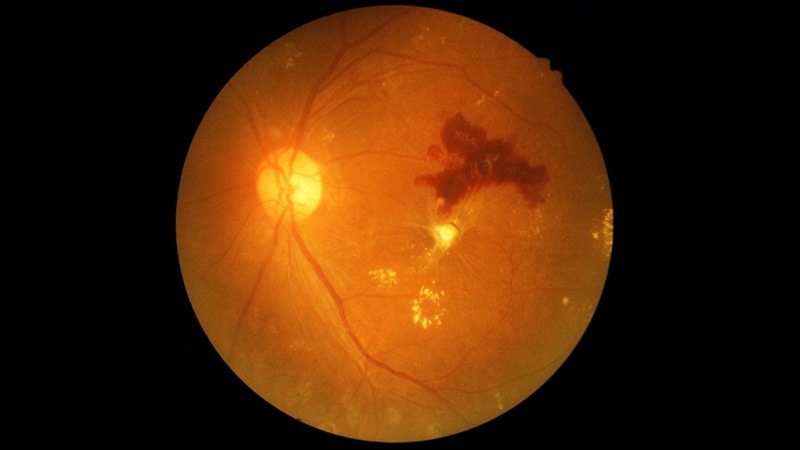
The initiative aims to accelerate research efforts to prevent and restore vision loss due to DRD, a term that includes diabetes-related retinopathy, macular edema, and retinal neurodegeneration. Its goals include the development of a new DRD staging system, identification of new endpoints and biomarkers, artificial intelligence-enhanced diagnostics, and ultimately new preventive approaches and treatments such as retinal regeneration or replacement.
“We brought together retina experts from all over the world, and one of the first things we decided to invest in was an update of the current Early Treatment Diabetic Retinopathy Study [ETDRS] staging system, which is 50 years old and unidimensional. It only looks the vascular side of the disease, and we now know that DRD is a disease of the entire retinal neurovascular unit,” Levine explained.
In fact, he noted, as many as 30% of people with diabetes who have normal-appearing retinas on photography already have demonstrable visual field defects. “So there needs to be a better way to stage. And once you have a better way of staging, you can create new indications for therapeutics development.”
Limitations of Current Treatments
While chronically elevated glucose levels raise the odds of developing DRD, everyone living with diabetes is at risk. One recent study showed that more than half the people who develop diabetes — type 1 or type 2 — in childhood develop DRD by a mean age of 26 years.
Current treatments include laser photocoagulation, anti-vascular endothelial growth factor (VEGF) injections, and surgery (vitrectomy). While these can preserve sight in many with late-stage DRD, they don’t work for everyone. Moreover, all involve some degree of risk, including scarring with laser treatments and infection with invasive eye injections, Jennifer Sun, MD, scientific co-director of the initiative, told Medscape Medical News.
“There’s a lot of room for new, more effective therapies that are nondestructive and noninvasive,” said Sun, who is also a professor of ophthalmology at Harvard Medical School, Cambridge, Massachusetts.
Moore herself underwent multiple laser treatments after being diagnosed with proliferative retinopathy in the mid-1980’s. At that point, she had lived with T1D for about 16 years.
“The laser preserved her central vision, so she continued to be able to read and to perform, but dancing requires peripheral vision and her visual fields were narrowed considerably. Over time, it got much worse, to the point where she really couldn’t safely cross a room without assistance. And ultimately, later in life, she was nearly blind,” Levine said.
“As a clinician, I would also love to see therapies that treat earlier stage disease, so that we’re not waiting for people to lose vision or be on the brink of losing vision before we treat them,” said Sun.
As well as looking for preventive strategies to help patients with diabetes maintain their vision, the initiative is also looking for therapies that work on a global perspective, that is “therapies that are readily accessible, affordable, and that can be safely administered, without the need for super-specialized care,” she added.
Public, Private, and Patients Together
Levine told Medscape Medical News that the initiative “has organized a public-private precompetitive consortium that enables industry to gain access to the data and research being developed by the initiative.” This consortium is based at the University of Michigan, with Harvard’s Joslin Diabetes Center as a lead academic partner. Funding comes from private philanthropy, including Breakthrough T1D, for core projects.
Boehringer Ingelheim joined the consortium in late 2024, followed by “numerous device manufacturers, biotech companies, and major pharmaceutical companies as members and project-specific supporters.”
Separately, the Entertainment Industry Foundation — which founded “Stand Up to Cancer” — and Variety magazine lend publicity support to the initiative.
“We’re positioning ourselves as an accelerator. We can’t do everything, but we’re doing what we can to attract investment, both of intellectual capital and dollars, to help accelerate the development of new therapeutics to eliminate vision loss,” Levine said.
While it doesn’t actually conduct clinical trials of new therapies, the initiative identifies needs, provides resource materials for researchers including an ocular biorepository and a retinal image database, sponsors a yearly symposium and workshops, and publishes papers to raise awareness of various aspects of DRD.
It also incorporates the patient voice. Patients and caregivers are invited to participate in a virtual meeting on February 12, 2026, to inform the FDA and other stakeholders about “experiences, perspectives, needs, and priorities” regarding drug development and regulatory decision-making.
The initiative also works with the longstanding DRCR Retina Network (formerly the Diabetic Retinopathy Clinical Research Network) funded by the National Institutes of Health (NIH) to support large multicenter clinical trials to validate biomarkers and endpoints that would be needed to accelerate product approvals.
Sun, who also co-leads the DRCR, explained, “We designed studies that we then brought to the DRCR investigator and coordinator group. We asked if they would partner with us to use the DRCR’s existing clinical trial infrastructure to perform them.”
Two upcoming studies for which the protocols were developed by the MTM Vision Initiative will be conducted by DRCR. One will examine functional endpoints in about 100 patients over the course of 1 year following anti-VEGF treatment for diabetic macular edema. The other is a larger and longer natural history study that will follow patients for over 4 years.
“I think these two studies will start to identify ways to characterize the retinas of people with diabetes, and start to build expectations of how we how we look at and describe patients’ visual function,” said Sun.
“Hopefully, these studies will provide ways to measure vision that will be more sensitive, more attuned to early changes, and that will help us efficiently find targets for new treatments.”
A ‘Different Angle’
Asked to comment on the initiative, Stephen J. Kim, MD, the Phyllis G. and William B. Snyder Chair and Retina Division Chief at Vanderbilt University, Nashville, Tennessee, told Medscape Medical News, “It a relatively new initiative that’s growing in popularity. They’re doing a good job of getting buy-in from experts to come to their meetings, to collaborate, and to have a shared vision.”
Compared to the traditional DRCR Network, the MTM Vision Initiative “has a different angle,” said Kim, who was an author of the American Academy of Ophthalmology’s 2025 Diabetic Retinopathy Preferred Practice Pattern. “It’s trying to incorporate new technologies, [and] new ways of diagnosing and defining the disease.”
This could help speed the time to new therapeutic discovery. “One of the main things” the initiative is doing is looking for “different biomarkers that can show us changes in the disease process,” Kim said.
“Right now, they’re just in the process of validating new markers for clinical trials. Once they start adopting or pushing one, there may be controversy,” he noted. “But it’s not controversial now.”
Moore’s Influence in Life and Beyond
Levine and Moore met in 1982, when she brought her mother into the emergency room at Mount Sinai Hospital in New York City one night when Levine happened to be on call. At the time, he was a cardiology fellow and founder of the institution’s cardiovascular disease prevention and rehabilitation program. They married the following year.
In 1985, Moore decided to accept the position as international chair of the organization then called the Juvenile Diabetes Foundation (JDF). “At that time, there weren’t a lot of celebrities willing to risk their careers by being public about their struggles with chronic disease,” Levine noted.
Moore was a dancer first, then an actor in two iconic American sitcoms — The Dick Van Dyke Show in the 1960s and The Mary Tyler Moore Show in the 1970s.
As a diabetes advocate, she helped raise billions of dollars for research funding. According to the site, “While Mary did not see a cure for diabetes in her lifetime, her legacy of leading efforts to ensure that future generations would not have to experience [its burdens] as she did, lives on through our work.”
Three years after Moore started as chair of the JDF, Levine left his cardiology practice to volunteer full-time for the Foundation, serving at various times as head of their government relations program and of their communications committee. He also helped develop their research strategy, and played a major role in securing bipartisan Congressional passage of funding for the Special Diabetes Program, which both advanced T1D research at the NIH and provided T2D treatment, education, and prevention programs for American Indian and Alaska Native populations. Today, Levine devotes nearly all his time to the MTM Vision Initiative.
The meetings and programs of the MTM Vision Initiative received educational and research grant support from a number of pharma, biotech, and device companies. Levine reported being occasionally provided with compensation and travel reimbursement for invited presentations for pharma collaborators.
Sun reported receiving research support from Adaptive Sensory Technology, Boehringer Ingelheim, F. Hoffmann-La Roche AG, Genentech, LKC Technologies, Konan, Novo Nordisk, Optovue, Inc., and Physical Sciences, Inc., and other relationships with Alcon, Boehringer Ingelheim, and Novo Nordisk. Kim disclosed having no relevant financial relationships.
Miriam E. Tucker is a freelance journalist based in the Washington, DC, area. She is a regular contributor to Medscape, with other work appearing in the Washington Post, NPR’s Shots blog, and Diatribe. She is on X @MiriamETucker and BlueSky @miriametucker.bsky.social.
<












Leave a Reply