April 13, 2026
5 min read
Key takeaways:
- Dopamine agonists target D2/D3 receptors, possibly increasing adverse events.
- Tavapadon targets D1/D5 receptors, reducing adverse event risks.
- Patient Global Impression of Change response improved with treatment.
Motor function improved with a favorable safety profile for patients in the early stages of Parkinson’s disease using tavapadon, according to a study published in JAMA Neurology.
Long-term use of oral levodopa, which is the current initial treatment, has been linked to motor complications, Angelo Antonini, MD, PhD, professor, Padova Neuroscience Center, University of Padova, and colleagues wrote.

According to the researchers, currently available dopamine agonists that target D2/D3 receptors may be less effective in achieving motor control than oral levodopa while increasing risks for adverse events .
“Tavapadon [AbbVie] is the first D1/D5 dopamine agonist showing efficacy in Parkinson therapy,” Antonini told Healio.

Angelo Antonini
“Most importantly, results from trials show it has a very low risk to induce impulse control disorders like gambling, compulsive shopping or hypersexuality compared to currently available agonists.”
Further, he added, the risk for dyskinesia and other involuntary movements appears lower with tavapadon, with a magnitude of motor benefits similar to other agonists.
TEMPO-1
The prospective, phase 3, multicenter, double-blind TEMPO-1 study included 529 patients (35.3% women; 97.2% white) aged 40 to 80 years (mean, 63.7 years) with early PD, defined as a duration of 3 years or fewer.
The cohort included 177 patients who received up to 5 mg of tavapadon, 177 who received up to 15 mg of tavapadon and 175 who received placebo, each once a day, for 27 weeks followed by a 4-week safety follow-up period.
A total of 43 patients in the 5 mg group, 59 in the 15 mg group and 27 in the placebo group discontinued participation, mostly due to adverse events or participant withdrawal.
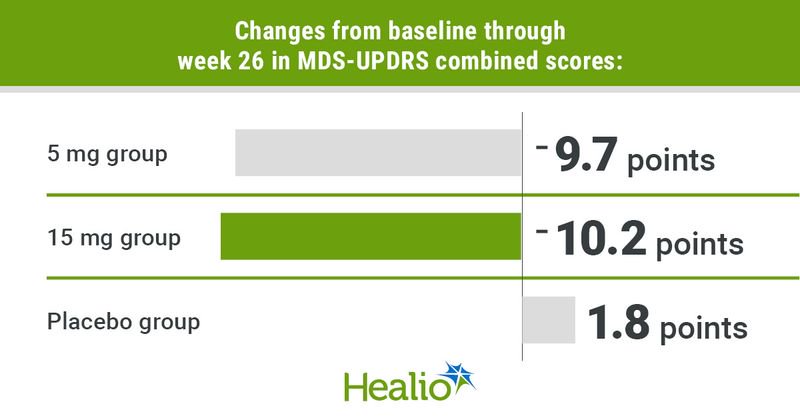
Least squares mean differences in Movement Disorder Society-Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) parts II and III scores from baseline to week 26 included –9.7 points for the 5 mg group, –10.2 points for the 15 mg group and 1.8 points for the placebo group. Improvements for the treatment groups emerged by week 5 and persisted through completion of treatment.
Specific least squares mean differences in MDS-UPDRS part II scores included –1.6 for the 5 mg group, –1.7 for the 15 mg group and 0.9 for the placebo group, with improvements emerging for the 15 mg group by week 5 and persisting through treatment completion.
Percentages of patients classified as Patient Global Impression of Change (PGIC) responders, with scores indicating “much improved” or “very much improved,” at week 26 included 45.5% of the 5 mg group, 44.4% of the 15 mg group and 12.2% of the placebo group.
Similarly, percentages of patients with any improvement in their PGIC scores at week 26 included 73.5% of the 5 mg group, 72.6% of the 15 mg group and 31.3% of the placebo group.
Percentages of patients with one or more adverse events included 80.2% of the 5 mg group and 78.5% of the 15 mg group, which the researchers called similar, and 57.1% of the placebo group.
Seven patients in the placebo group and 64 in the treatment groups discontinued treatment because of adverse events. Once titration was over, risks for discontinuation fell. Adverse events were mostly called mild to moderate and not serious.
The treatment groups experienced more nausea, headache and dizziness than the placebo group, the researchers said, with the most common adverse events happening less often during maintenance, compared with titration.
Rates of somnolence included 3.4% for the 5 mg group, 2.8% for the 15 mg group and 3.4% for the placebo group, which the researchers called similar.
Three patients in the treatment groups experienced mild to moderate adverse events suggesting impulse control disorders (ICDs) during maintenance.
Least square mean changes in total Questionnaire for Impulsive-Compulsive Disorders in Parkinson Disease Rating Scale (QUIP-RS) from baseline to week 26 included -2.2 for the 5 mg group, –2.4 for the 15 mg group and –2.1 for the placebo group, which the researchers called similar.
Eleven patients in the 15 mg group experienced hallucinations, but only one discontinued treatment. The other events were all called mild or moderate. Rates of peripheral edema included 2.3% for the 5 mg group and 0.6% for the 15 mg group, with no cases in the placebo group.
Additionally, patients in the treatment groups experienced comparable decreases in mean systolic and diastolic blood pressure during titration that gradually fell during treatment. Patients in the placebo group did not have any changes in their blood pressure.
Next steps
Based on these findings, Antonini and colleagues said that tavapadon led to clinically meaningful and significant improvements in symptoms of patients with PD.
“Neurologists are currently very concerned about using traditional dopamine agonists, but the safety profile of tavapadon with low risk of impulse control disorders should reassure many of them about the benefits of this drug class,” Antonini said. “Tavapadon may offer a safe complementary treatment to levodopa and should expand the therapeutic portfolios for Parkinson’s patients.”
The researchers called for long-term safety and efficacy studies of tavapadon among patients with early PD.
“All results are, of course, limited to few months of treatment and need to be confirmed in longer observational studies,” Antonini told Healio.
“Especially the lower risk of dyskinesia, if confirmed by longer studies, may represent a significant advancement, as this may delay or reduce severity of a disease milestone,” he added. “Lower dyskinesia means also potentially reduced need to progress to invasive device therapies like deep brain stimulation or infusions
For more information:
Angelo Antonini, MD, PhD, can be reached at angelo.antonini@unipd.it.
<












Leave a Reply