Under a fluorescence microscope at Northwestern University, a sphere of human spinal cord tissue — about 3 mm across, roughly the diameter of a mouse spinal cord — erupted with green light. It had been stained with a calcium-sensitive dye that lights up living neurons and their extending fibers. This green light provided unmistakable visual proof of neurites streaming outward from the tissue’s injured surface and into a synthetic gel, where they were growing in organized, parallel structures.
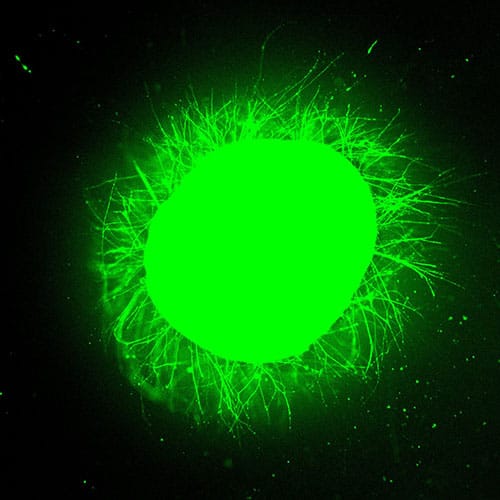
That sphere of spinal cord tissue was a spinal cord organoid — a three-dimensional, miniaturized tissue model grown in a dish from human induced pluripotent stem cells (iPSCs) to recapitulate key features of an actual organ. This one had been differentiated over 24 weeks into a range of spinal cord cell types: neurons, astrocytes, oligodendrocyte progenitor cells, and Schwann cells.
The Northwestern team also incorporated microglia — the brain and spinal cord’s resident immune cells — by coaxing iPSC-derived progenitors to infiltrate the organoid. The result was an immune-competent model capable of mounting an inflammatory response to injury.
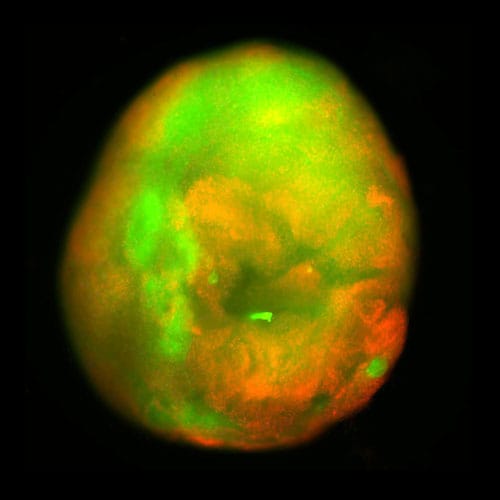
Then, to make the organoid useful, the team injured it — either slicing it with a scalpel to model surgical hemisection or crushing it with a mechanical impactor to deliver the kind of compressive contusion that causes most real-world spinal cord injuries.
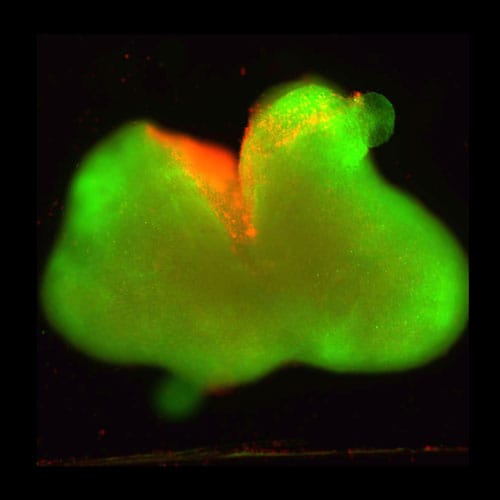
Twenty-four hours later, they treated the lesion with an experimental “supramolecular” therapy: a liquid solution of synthetic peptide molecules that, on contact with biological tissue, spontaneously assemble into a regenerative scaffold filling the injury site. Because the structure is held together by weak, reversible forces rather than rigid chemical bonds, the molecules retain freedom to move even after assembly. That motion turns out to be critical.
In textbook-like fashion, the untreated organoids responded just as an injured spinal cord would: neurons died, and over weeks a dense glial scar formed — the same barrier of reactive astrocytes that blocks regeneration in patients. No axons regrew on their own. This was critical validation: the organoid wasn’t just damaged tissue, but tissue that recapitulated the actual pathology of spinal cord injury.

The organoids treated with the experimental therapy told a different story.
“We saw this spectacular growth of axons from the cells that were on the surface of the organoid as soon as they became in contact with our synthetic filaments,” said Samuel I. Stupp, PhD, director of Northwestern’s Center for Regenerative Nanomedicine and senior author on the study. “It was really spectacular to see.”
Recently published in Nature Biomedical Engineering, the study represents the first reported use of a deliberately injured human spinal cord organoid to test a candidate regenerative therapy. It builds directly on work Stupp’s team published in Science in 2021, when they showed that a single injection of the same synthetic gel could reverse paralysis in mice. They built the organoid model to confront a stubborn reality: therapies that work in rodents routinely fail in people.
The organoid offers a human-tissue checkpoint in that pipeline. The question is how much it can reveal.
How the ‘Dancing Molecules’ Work
The therapy for regeneration used in the study is a synthetic compound Stupp’s lab originally formulated in 2001. The molecules — peptide amphiphiles — self-assemble into nanofibers that display a bioactive peptide signal derived from laminin, a key protein in the body’s natural extracellular matrix. In solution, the nanofibers stay dissolved because their electrical charges repel each other.
On contact with the body’s salt-rich fluids, the electrical repulsions between nanofibers diminish and they collapse into a scaffold resembling natural extracellular matrix. The resulting scaffold fills the lesion cavity and, as Stupp put it, “hugs the cells.”
But the scaffold’s physical presence is only half the story. Because the structure is supramolecular — assembled through weak, reversible interactions rather than permanent covalent bonds — the molecules retain freedom to move within the assembled fibers. That internal “dance” determines the therapeutic potency of the scaffold.
The most dynamic version, IKVAV PA2, drove neurite growth from organoid surfaces at 5.01 µm/h — roughly 120 µm/d — enough to span a small lesion gap within weeks. It was more than double the rate of a slower-moving variant and over three times that of a nonbioactive control.
“Only the systems with enhanced motion could reverse the paralysis,” Stupp said of their earlier animal work.
Treated organoids showed dramatically less scarring than untreated ones and surviving neurons had extended long projections into the synthetic gel. In the immune-competent organoids, neurofilament staining — a marker specific to nerve fibers — confirmed the outgrowth was genuine axons. The long axonal extensions were consistent across both organoid types.
The immune-competent model revealed an additional dimension. Bulk RNA sequencing showed significant downregulation of pro-inflammatory genes including TNF-alpha and interleukin-1-beta, and the treatment reduced chondroitin sulfate proteoglycans, which inhibit axons from regrowing. Levels were reduced to nearly those of an uninjured model.
The scaffold was working on two fronts simultaneously: suppressing the physical scar and clearing the chemical blockade.
Unlike cell-based approaches, the fully synthetic scaffold is biodegradable. Once its regenerative work is complete, the material breaks down into nutrients the spinal cord absorbs.
What the Organoid Can’t Tell You

Michael Fehlings, MD, PhD, a neurosurgeon at the University of Toronto and editor-in-chief of the journal Spinal Cord, led the landmark STASCIS trial that established early surgical decompression as the global standard of care. He situates Stupp’s work within a broader turning point.
“We are probably in the early phases of an era of regenerative neuroscience,” Fehlings said. After years of patients traveling overseas for unproven “voodoo cocktails,” as he put it, legitimate breakthroughs are finally emerging from labs in the US, Canada, and Europe. These therapies are rigorous enough to advance into real clinical trials. The organoid, in his view, is part of that maturation: not a cure, but a checkpoint on the path toward one.
He characterized the organoid as occupying a middle ground in translational research. “It’s sort of halfway between a cell culture situation and an in vivo situation.” Because the organoid is grown in a dish, it’s “separated from the immunological response in vivo, and it’s separated from the vascularization of the tissue,” he added.

Pengzhe (Paul) Lu, PhD, a neuroscientist at the University of California San Diego whose own work has demonstrated functional recovery in severe chronic spinal cord injury using neural stem cell grafts combined with rehabilitation, raised an even more fundamental concern: the organoid lacks white matter.
In a living spinal cord, roughly two thirds of the tissue consists not of neurons, but of myelinated axon tracts — the long-distance wiring that carries signals between brain and body. Because the organoid is isolated, with no connection to a brain or peripheral nervous system, those tracts simply don’t exist. “That’s a big difference between the real model, in vivo model, and the in vitro model,” Lu said.
But the precise role of glial scars in spinal cord injury remains debated. Lu, whose own neural stem cell grafts extend axons directly through the scar without difficulty, falls on the “beneficial” side of the controversy. “The glial scar is not a barrier for them,” he said of his transplanted cells. Yet others argue the scar blocks regeneration. Stupp’s data support the latter view, at least for synthetic, cell-free approaches.
Still, in one critical respect, the organoid did behave like the real thing. The presence of glial scarring and chondroitin sulfate proteoglycans — and the absence of any spontaneous regrowth — suggested the organoid behaves like adult spinal cord tissue, not the neonatal tissue that can regenerate on its own. That distinction matters: any therapy that works here faces the same hostile environment it would encounter in a patient.
What Comes Next
For Fehlings, the organoid’s most promising future may lie in testing therapies far more complex than any single scaffold or cell graft alone. He envisions a staged approach: first, a bioactive scaffold like Stupp’s to bridge the lesion cavity and reshape the hostile microenvironment; then, engineered neural stem cells to replace lost neurons and rebuild functional circuitry.
“I think that would be quite an exciting therapy,” he said. In fact, Fehlings noted he is “quite intrigued” by the prospect of combining his own neural stem cell research with Stupp’s scaffold in the organoid.
Lu’s own work supports the combinatorial approach. In a 2022 study, his team showed that neural progenitor cell grafts combined with intensive rehabilitation produced significant functional recovery in rats with severe chronic cervical injuries. But neither treatment alone was effective. Rehabilitation appeared to enhance axon regeneration into the grafts, suggesting it’s necessary for full functionality during the process.
Stupp’s lab is now pushing toward human trials for acute spinal cord injury and has been in discussions with the FDA, all while developing a more advanced version of the therapy targeting chronic injuries, which represents a more intractable patient population.
In preclinical models, the team recreates chronic injury by waiting for 2 months post-injury in rats, so the same fluid-filled cavity and mature glial scar seen in human patients emerges. Stupp said he hopes to report results from this work later in 2026.
Lu, who has worked in the field for three decades, offered a historic perspective: early theories blamed inhibitory molecules in myelin; later work pointed to the glial scar. Both proved incomplete. “We’ve gone up and down,” he said. The field has shifted toward understanding why adult neurons shut off their growth programs entirely — and whether those programs can be reactivated.
Stupp, who receives emails from paralyzed patients around the world nearly every day, offered a direct message: “They should be hopeful that there’s something on the horizon for them.”
“The science is valid,” Fehlings said. “This is from a very reputable group, and the work is of high quality.” But he urged patients and families to calibrate their expectations: the field is advancing, but carefully. “We’ll see where this goes.”
Stupp reported that he and several co-authors were co-inventors on a patent application related to this work filed by Northwestern University (US patent application number 18/735,858, filed on June 6, 2024). Fehlings reported a past professional collaboration with Stupp’s laboratory on a paper evaluating an earlier version of the bioactive peptide, and noted he recently served as a visiting professor at Northwestern University. Lu did not report any relevant financial conflicts. Disclosure information for study authors is available in the original study publication.
<










Leave a Reply