April 14, 2026
2 min read
Key takeaways:
- David F. Chang, MD, called for clearer regulations on multi-use surgical items.
- Transitioning to reusable phaco cassettes has the potential to curb the environmental impact of cataract surgery.
WASHINGTON — Clearer regulations on reusable items may help reduce surgical waste, according to a speaker at American Society of Cataract and Refractive Surgery meeting.
During the Richard L. Lindstrom Lecture, David F. Chang, MD, said sustainability issues in ophthalmology have made it clear that more multi-use items are needed to reduce operating room waste. However, regulatory classifications make it difficult for surgeons to get their hands on multi-use items.

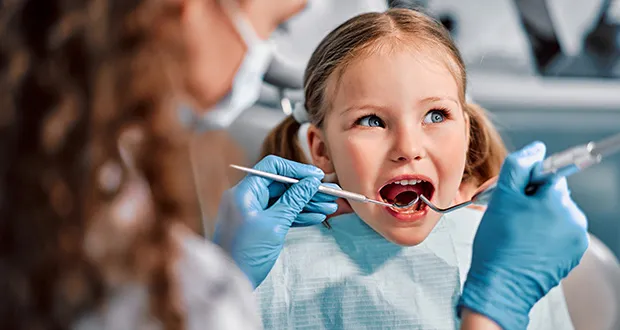
At the ASCRS meeting, David F. Chang, MD, (center) was honored with the Richard L. Lindstrom Medal by Cathleen McCabe, MD, (left) and Richard L. Lindstrom, MD, (right). Image: Eamon N. Dreisbach | Healio
“For a manufacturer to get a label for [reuse] five times, it has to demonstrate to the FDA it’s safe to reuse 10 times,” Chang, who was awarded this year’s Lindstrom Medal, said. “If you don’t do that, the default is it’s labeled single-use.”
When surveying surgeons about sustainability issues, Chang said he found that about 75% of responders believed that they should be able to reuse phacoemulsification cassettes and irrigation bottles on multiple patients. Using these items for multiple patients is a much more common practice in Asia. Chang said that discrepancy is all about regulations. Hospital networks like the Aravind Eye Care System in India use one cassette and throw it away at the end of the day, he said.
To get a clearer picture of how single-use and multi-use practices compare in terms of endophthalmitis rates, Chang said he and colleagues took 10 years of data on cataract surgery from the Aravind Eye Care System and found a rate of less than 0.01% across more than 1 million cases. In the U.S. over the same 10-year span, the rate was 0.06% across about 9.7 million cases. They also published a paper on microbiology, taking samples from phaco cassettes and tubing, and the cultures were negative, Chang said.

Chang said the U.S. and other Western countries account for more than 60% of global phaco cassette sales. He believes that by transitioning to reusable cassettes, there could be savings on the manufacturing side to lower surgeon facility costs and make it more efficient while maintaining manufacturing margins.
As the lead author on an EyeSustain position paper, Chang also called for the phaco machine industry to prioritize development of multi-use phaco cassettes, tubing and other supplies and for the FDA and other regulatory agencies to facilitate and expedite review of multi-use products.
“We want the FDA to have a better classification system,” Chang said. “Make it clear what’s a requirement versus a suggestion. CMS, at least in ophthalmology, needs to go back to where it was in 2009 when we had the discretion to use our judgment for what we use off-label.”
<












Leave a Reply