Preventive treatment with fremanezumab was associated with significantly reduced migraine frequency and headache severity compared with placebo in children and adolescents with episodic migraine, new data showed.
In the phase 3 SPACE trial, fremanezumab 120 mg and 220 mg monthly led to greater reductions in monthly migraine days and higher response rates among patients aged 6-17 years than placebo. Treatment for 3 months was also associated with fewer days of moderate-to-severe headache and a decreased use of acute migraine medication.
Fremanezumab, a subcutaneous calcitonin gene-related peptide (CGRP) inhibitor, is approved in the US as a preventive migraine treatment in adults for both monthly and quarterly dosing. Last year, the FDA expanded the indication to include pediatric patients with episodic migraine for monthly dosing, a decision based on early results from the SPACE trial.
The trial is the first phase 3 study to demonstrate a significant reduction in monthly migraine and headache days with fremanezumab for episodic migraine in this patient population.
The data “demonstrate clinically meaningful benefits of CGRP-targeted therapy in pediatric migraine and help address a longstanding gap in evidence for preventive treatment in this population,” lead investigator Andrew D. Hershey, MD, PhD, professor of neurology, Cincinnati Children’s Hospital Medical Center, Cincinnati, Ohio, and colleagues wrote.
The full results were published online on January 14 in The New England Journal of Medicine.
Limited Options for Children
Migraine is a common neurologic disorder in children and adolescents, affecting an estimated 11% of this population. It contributes to missed school days, poor academic performance, and a decreased quality of life.
Although lifestyle modifications and acute treatments are commonly used, preventive treatment options for pediatric migraine, such as topiramate, propranolol, and amitriptyline, have limited efficacy and are associated with adverse effects (AEs). CGRP antibody therapies are effective in adults, but data from randomized controlled trials were limited in pediatric patients.
SPACE was an international, multicenter, randomized, double-blind, placebo-controlled trial that included 237 participants, with a full analysis set of 234 patients aged 6-17 years with an episodic migraine diagnosis of at least 6 months, with or without aura.
Patients were required to have fewer than 14 headache days per month during the prescreening period. Up to 30% of participants were allowed to continue stable concomitant therapy; patients had to be on a fixed dosage of no more than two medications at least 2 months prior.
Participants received either placebo (n = 112) or monthly subcutaneous injections of fremanezumab or placebo for 3 months, with weight-based dosing of 120 mg for patients weighing less than 45 kg (n = 36) and 225 mg for those weighing 45 kg or more (n = 87).
Participants were allowed to continue the use of migraine-specific medications for acute treatment as needed.
Fewer Migraine Days
The primary endpoint was the change from baseline (28-day period) in the average number of migraine days per month during the 12-week treatment period.
The treatment group vs placebo group reported a significantly greater decrease in monthly migraine days (2.5 days vs 1.4 days), with a between-group difference of 1.1 days (P = .02).
Nearly half of patients treated with fremanezumab (47.2%) achieved a 50% or greater reduction in monthly migraine days compared with 27% of patients who received placebo (P = .002).
Fremanezumab was also associated with a decrease in the number of days with headache of at least moderate severity and fewer days of acute migraine medication use.
“These improvements are particularly meaningful given the impact migraine can have on school attendance and daily functioning in children and adolescents,” the investigators wrote.
Safety Findings
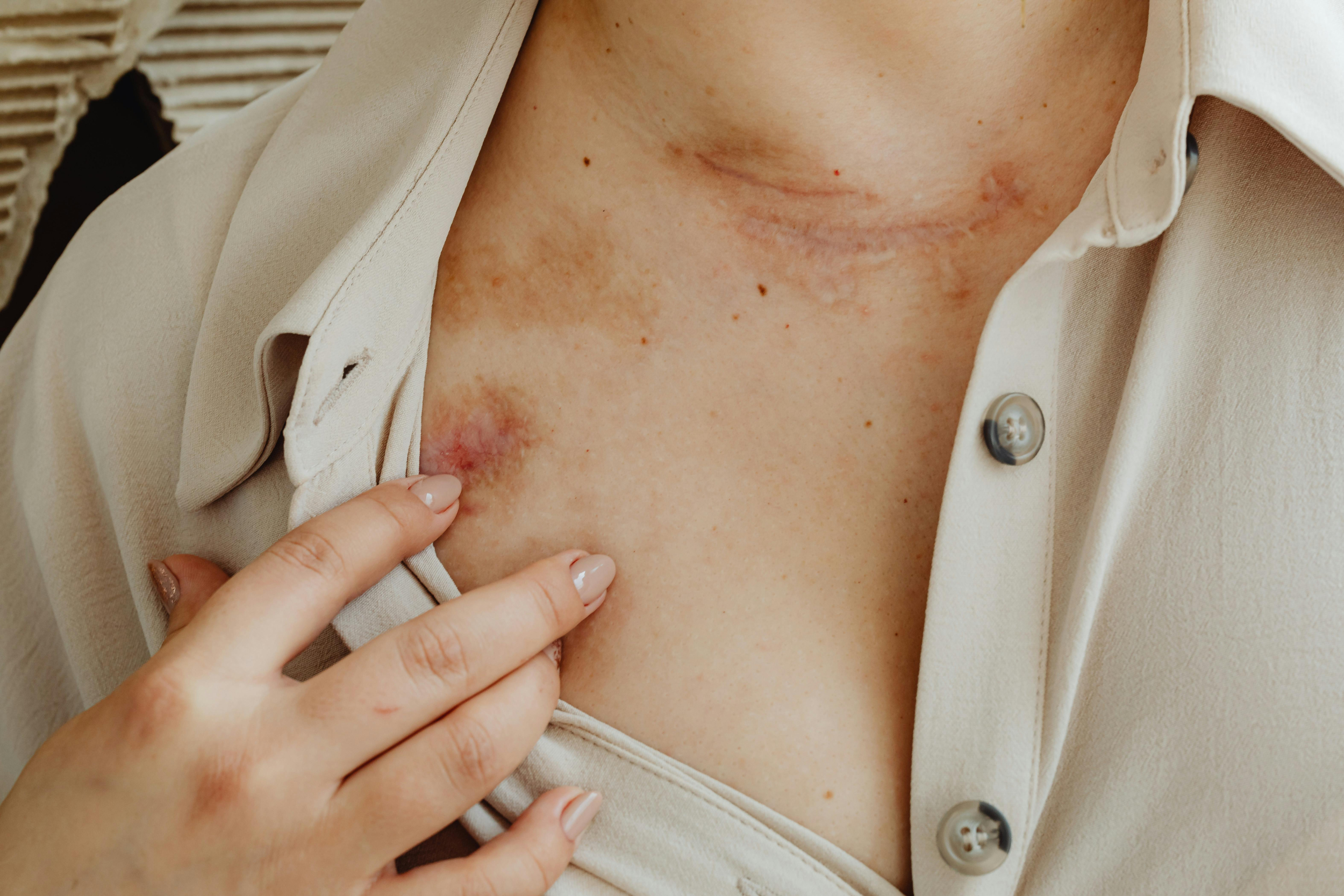
AEs were generally mild to moderate and occurred at similar rates in both groups. Injection-site erythema was the most frequently reported AE (9.8% with fremanezumab vs 5.4% with placebo).
In the fremanezumab group, two participants reported serious AEs, including hepatitis secondary to infectious mononucleosis and migraine exacerbation requiring hospitalization. In the placebo group, three participants reported serious AEs, including thrombocytopenia, hemiparesis, and migraine.
No new safety signals were identified during the study period.
“Except for injection-site reactions, no safety concerns were identified, and the overall safety profile was consistent with that observed in adults,” the investigators wrote.
Study limitations included the 3-month double-blind treatment period and the need for longer-term follow-up through open-label extensions to further assess the long-term safety and lasting effects in this patient population.
The study was funded by Teva Pharmaceuticals. Hershey reports receiving personal fees and nonfinancial support from Teva during the conduct of the study, as well as personal fees from AbbVie, Biohaven, Eli Lilly, and Lundbeck outside the submitted work. Several other study authors also reported financial relationships with Teva and other pharmaceutical companies.
<












Leave a Reply