April 24, 2026
3 min read
Key takeaways:
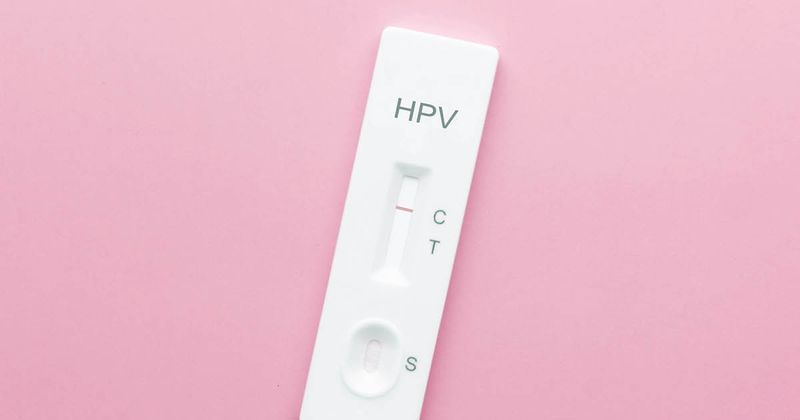
- ACOG’s guidance for the first time includes the option to self-collect for primary high-risk HPV screening.
- The guidance “is a huge step forward” for expanding access to screening, an expert said.
ACOG issued updated guidelines on screening for cervical cancer that align with recent recommendations from the Health Resources and Services Administration’s Women’s Preventive Services Initiative.
The guidance for the first time includes the option for patients aged 30 to 65 years at average risk for cervical cancer to collect their own samples with FDA-approved testing kits for primary high-risk HPV (hrHPV) screening every 3 years, according to an ACOG press release.

The organization advised that clinicians should only offer the option of self-collection “if patients prefer this method and if appropriate clinical infrastructure and oversight for notification, documentation and referral for follow-up as indicated are in place.”
Primary hrHPV testing every 5 years is preferred for women aged 30 to 65 years, the recommendations published in Obstetrics & Gynecology said, while the guidance offers the option of co-testing with hrHPV and cervical cytology.
Cervical cytology alone in this age group “should be used only in settings in which primary hrHPV testing or co-testing is not available or if, after counseling, the patient chooses cervical cytology alone,” according to the recommendations.
Women aged 21 to 29 years should receive screening with cervical cytology every 3 years, the guidance states, while women older than 65 years who are either at high risk or have not received adequate prior screening, “defined as three consecutive negative cytology results or two consecutive negative co-testing results within 10 years before stopping screening, with the most recent test occurring within 3 years for cytology alone or 5 years if co-testing is used,” should continue to be screened.
“Too many people in the United States are needlessly suffering from cervical cancer. Underscreening — and lack of screening — is the most significant contributor to the development of this largely preventable illness,” ACOG President Steven J. Fleischman, MD, MBA, FACOG, said in the release. “All patients deserve access to screening options grounded in the best available science, and it is our goal to ensure that clinicians have the clear, evidence-based guidance they need to make that happen.”
According to guideline author Christopher Zahn, MD, FACOG, ACOG’s chief of clinical practice, women in underserved communities have higher cervical cancer incidence and mortality rates, “and closing the gap requires screening to be appropriate, evidence-based and accessible. These updated recommendations, including patient-collected hrHPV testing, give clinicians new tools to reach patients who have historically faced barriers to screening — but this expanded access must be paired with appropriate counseling and the necessary infrastructure.”
Andrew M.D. Wolf, MD, FACP, Professor Emeritus of Medicine at University of Virgina School of Medicine and chair of the American Cancer Society Screening Guidelines Development Group, told Healio that inclusion of the option for self-collected testing “is a huge step forward to expand access to screening and save more lives. Most self-collection devices currently available are only approved for use in the clinical setting, meaning patients collect the sample while in the office. This is still a big step forward, since many primary care clinicians defer cervical cancer screening due to lack of time during the office visit — self testing can address this barrier.”
Wolf added that many women “will prefer self-testing over clinician exams for comfort reasons. Currently, only the Teal Wand [Teal Health] is approved for at-home collection, but there will be more options down the road. Clinicians and patients should also understand that if the self-collected HPV test is positive, the patient will need to return for a clinician-collected cytology (pap) test.”
Wolf underlined that cervical cancer “is almost completely preventable by HPV vaccination. Clinicians should be vaccinating all children between the ages of 9 and 12, and definitely by age 26.”
*Editor’s note: Wolf’s interview reflects his own views and opinions, not his affiliations or institutions.
For more information:
Andrew M.D. Wolf, MD, FACP, can be reached at primarycare@healio.com.
<













Leave a Reply