Existing treatments for other conditions show potential for use in managing type 1 diabetes, a recent scientific conference heard
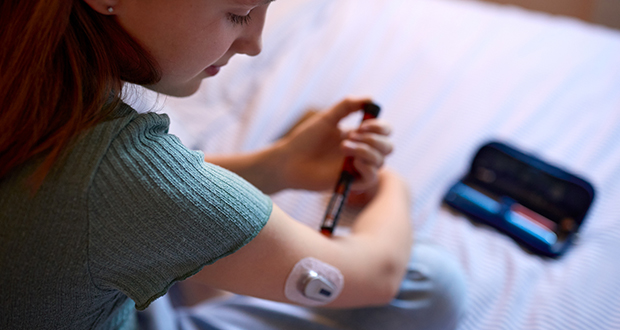
The number of people living with type 1 diabetes (T1D) is set to grow substantially in the coming years. A study published earlier this year estimates that currently 9.5 million people worldwide live with the condition, with this number set to increase to 14.7 million by 2040.
While the treatment landscape for T1D is evolving rapidly, identifying people with the disease remains a challenge. The study estimates that around 4.1 million ‘missing people’ would still be alive in 2025 if they hadn’t died prematurely from poor T1D care.
This includes an estimated 669,000 who were never diagnosed with the condition.
“Our first priority should be to limit the rise in the number of people who are dying prematurely due to lack of diagnosis or suboptimal diabetes care,” said Dr Fei Wang, a consultant with US-based research and advocacy organisation Breakthrough T1D.
“The number of these ‘missing people’ is expected to reach a staggering 6.7 million by 2040, yet millions of these lives could be saved with existing treatments and technologies.”
This analysis was presented at the annual meeting of the European Association for the Study of Diabetes (EASD), held in Vienna, Austria, earlier this year.
Currently 9.5 million people worldwide live with type 1 diabetes
The event heard about new discoveries that may enhance the treatment of T1D, including research looking at an existing immunosuppressant medication used to help prevent or treat acute rejection after organ transplants.
Anti-thymocyte globulin (ATG) is made of antibodies that target T lymphocytes – cells of the immune system that can attack the body’s own tissues (as happens in T1D).
This clinical trial, which was published in the Lancet, found that a much lower dose than previously thought of ATG is safe and effective in preventing progression of T1D in younger people.
“The MELD-ATG trial showed that an adaptive trial design for testing an intervention aiming to arrest the loss of functional beta-cell mass in young people with recent-onset clinical, stage 3 T1D successfully identified a minimum effective dose of 0·5mg/kg ATG,” its authors said.
“In addition, it allowed confirmation of previous efficacy and safety findings of 2·5mg/kg ATG, including in children as young as five years of age.
“Low dose ATG is a safe and effective intervention for arresting or at least delaying progression of T1D… This adaptive trial design can be considered for further exploration of novel therapies in T1D, and even for other fields.”
The authors add that the MELD-ATG results underscore the need to perform immune modulatory intervention studies directly in young individuals with T1D, in contrast to current drug development strategies that, driven by regulatory guidance, typically target adults first.
“Especially in the youngest age group, the 0·5mg/kg dose was effective with a good safety profile and would be the recommended dose for treatment,” they said.
“Of interest, being able to limit the administration of ATG to 0·5mg/kg would also mean only needing one infusion on one day, instead of the two days of infusion with the previously studied 2·5mg/kg.”
The EASD meeting also heard results of a follow-up trial investigating whether a daily pill could slow the onset of T1D.
In 2023, the groundbreaking Australian BANDIT trial reported that a daily pill of baricitinib, commonly prescribed for rheumatoid arthritis and alopecia, could safely preserve the body’s own insulin production and slow the progression of T1D in people recently diagnosed with the condition.
The follow-up of the study recently revealed that once baricitinib treatment was stopped, participants’ diabetes progressed – they produced less insulin and had less stable blood sugar levels, that were not significantly different to those on placebo.
“Among the promising agents shown to preserve beta cell function in T1D, baricitinib stands out because it can be taken orally, is well-tolerated, including by young children, and is clearly efficacious”, said author Dr Michalea Waibel from St Vincent’s Institute of Medical Research, Fitzroy, Australia.
“These latest data support our previous clinical trial data by showing that the therapeutic effect is lost when baricitinib is stopped and justify further trials to determine if treatment benefit can be sustained over many years on treatment and if treating earlier stages of disease can prevent or delay clinical diagnosis.”
Another study highlighted the potential effects of verapamil in slowing the progression of T1D.
Verapamil is a well-known drug for high blood pressure and other heart diseases and has been used in adults and children for more than five decades.
Previous studies have shown that certain types of overexpression of proteins can lead to beta cell death, and verapamil, a calcium channel blocker, can prevent this overexpression and thus potentially preserve beta-cell function.
The Ver-A-T1D trial investigated whether verapamil SR (360mg daily) could preserve beta-cell function in adults with newly diagnosed T1D.
This randomised, placebo-controlled study recruited 136 participants across 21 sites in six European countries.
The primary endpoint measured stimulated C-peptide response (a marker of beta cell function) during a two-hour mixed meal tolerance test at 12 months.
Whilst the study narrowly missed statistical significance, it demonstrated encouraging trends towards beta-cell preservation with verapamil treatment.
“The unique non-immunosuppressive mechanism, the established safety profile and cost-effective delivery make verapamil still an attractive option for combination with immune modulating interventions for beta-cell preservation in type 1 diabetes,” said lead author Prof. Thomas R. Pieber of Medical University of Graz, Austria.
“The next important step will be to analyse the 24-month follow-up data of Ver-A-T1D to find out how well insulin-producing capacity will be preserved in our patients without treatment.
“More importantly, in the Ver-A-Long extension study, we will follow patients who did continue with verapamil treatment after the Ver-A-T1D trial. This will give us very important information about long-term preservation of beta cell function over three years when verapamil is continuously used in newly diagnosed people with type 1 diabetes.” ![]()
<







Leave a Reply